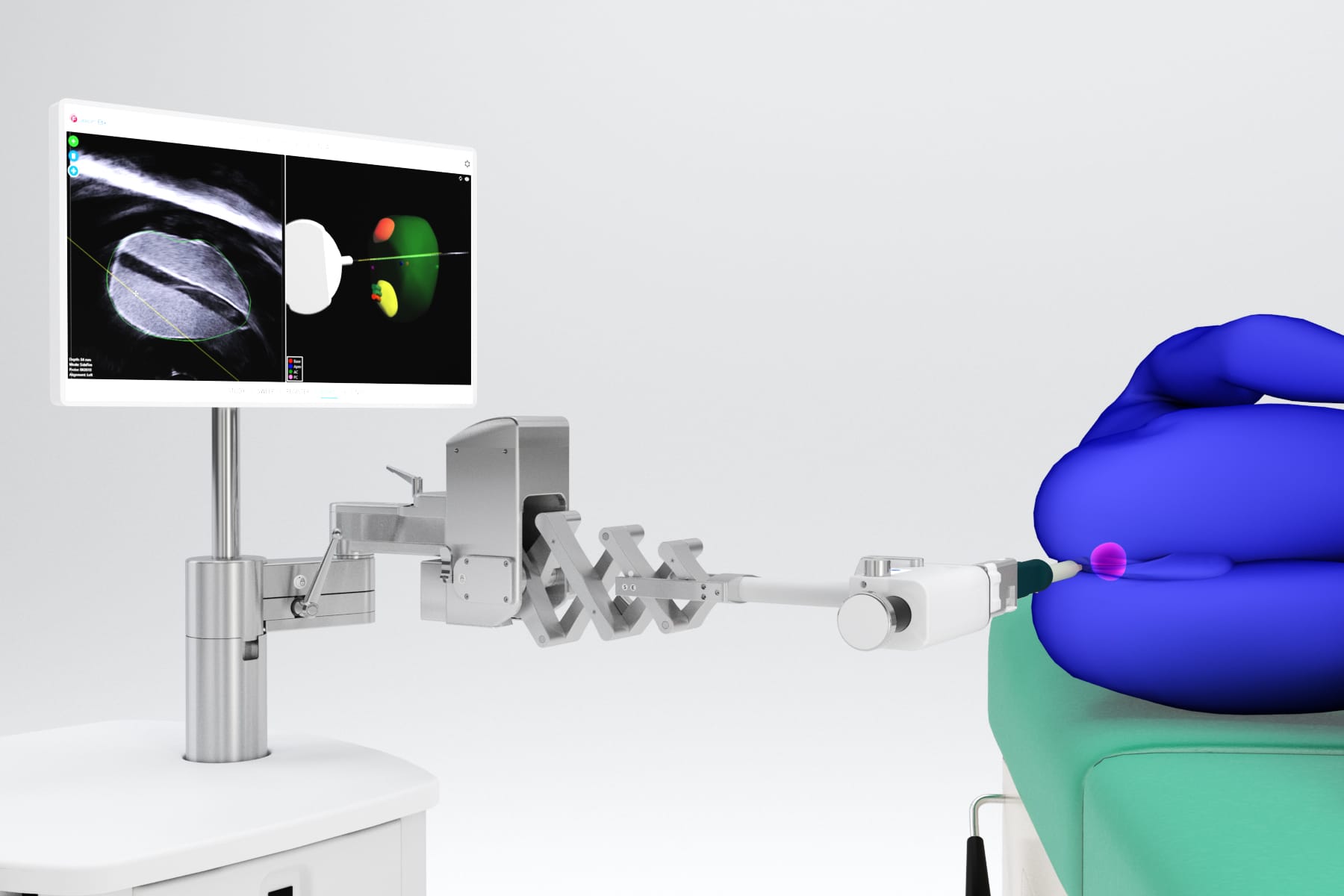
What is ‘tracking’ in fusion biopsy?
In the field of prostate fusion biopsy, tracking refers to “the ability to visualize (in real time) the TRUS-probe position and [projected] needle advancement in 3D space throughout the procedure”.2 Because the location of the ultrasound probe and projected needle path are constantly tracked, the operator can navigate to image slices of interest under complete visualization rather than through a traditional “blind” sampling of the prostate. Tracking the projected needle path also allows the physician to monitor the precise location of a biopsy sample for treatments like active surveillance.
Why is the type of tracking important to a fusion biopsy procedure?
Tracking is of central importance to the fusion biopsy procedure because the precise targeting enabled by MRI-US fusion relies on knowing exactly where the ultrasound probe is in relation to the prostate at all times. If the location of the ultrasound probe is not accurately tracked, the overall accuracy of the fusion biopsy procedure is put into jeopardy. The mechanical and logistical processes of different tracking techniques vary and — as a result — can affect not only the accuracy of the procedure, but also patient comfort, urology practice operations and finances, as well as the general ease-of-use of the device.
Semi-robotic vs. Electromagnetic solutions
The type of tracking used during a fusion biopsy procedure generally falls under one of two categories: semi-robotic tracking or electromagnetic tracking.
Electromagnetic (EM) Tracking
Technique
Electromagnetic tracking follows Faraday’s Principle, which states that current can be generated in a solenoid (a coil of wire capable of converting electrical energy to mechanical energy) when placed in a continuously changing magnetic field. Under the electromagnetic tracking method, a small sensor is attached to the freehand TRUS-probe. A magnetic field generator is placed above the patient (usually attached to the operating table) which generates a sequentially changing magnetic field. During the procedure, the sensor on the ultrasound probe generates a current in response to the changing magnetic field created by the field generator. The current produced by the sensor is relayed to the fusion device’s computer, which is used to determine the location of the probe with respect to the electromagnetic field. This information is then used to guide the physician towards regions of interest predefined on MRI.2
Pros and Cons
This technique offers a more familiar approach in hardware design as the clinician will already know how to use the ultrasound probe in a freehand manner. Additionally, the freehand method allows for multiple degrees of access during the procedure and feels intuitive to use.
However, the use of the freehand method in electromagnetic tracking also allows for the introduction of human error. Biopsy procedures that use a freehand technique are more likely to cause prostate deformation than their semi-robotic solution counterparts. When the shape of the prostate is deformed, registration is no longer reliable because the registered contour does not line up with the prostate in live ultrasound. This leads to inaccuracies. Additionally, maintaining consistent pressure on the prostate is more difficult to achieve with a lack of mechanical support.3 To complicate things further, the operating physician must keep their hands on the probe at all times during the procedure, limiting mobility. With the physician’s hands full, an additional assistant is often needed, which may require the dedication of additional time and money on part of the practice.
Furthermore, electromagnetic tracking requires the added hardware and software components of both a magnetic field generator and disposable ultrasound probe sensors. During biopsy, the field generator must be moved and re-calibrated for each patient, adding to procedure length. The field generator is susceptible to interference from other metallic objects in the workspace which can distort the machine’s signal, possibly resulting in inaccurate tracking of the entire procedure.4 These devices bring other cost-based and logistical issues to a urological practice, requiring continued purchasing of disposable sensors and dedicated training time to gain familiarity with the magnetic field generator.
Semi-Robotic Tracking
Technique
Semi-robotic tracking uses a robotic arm or a mechanical stepper to track the location of the TRUS probe through direct attachment of the probe to the device. The position of the probe with respect to the prostate in 3D space is encoded and automatically sent to the computer through angle sensors located in the robotic arm joints. As the ultrasound probe is manually rotated, the sensors detect movements in each of the joints and translate this information into a position in 3D space.2
Pros and Cons
With the steady support offered by its mechanical arm, the semi-robotic tracking technique increases accuracy by addressing human error that can be introduced via the freehand method. Rothwax et. al state that robotic tracking technology “offers superior accuracy and the ability to completely immobilize the probe from target acquisition to firing of the needle”.5 The mechanical arm provides consistent pressure on the prostate, allowing for maximum accuracy of registration and consistent coupling. Additionally, it enables the physician to take hands-free pauses, reducing or eliminating the need for an assistant. Alternatively, the assistant can set up the biopsy for the clinician to perform, reducing the time spent by the clinician during the procedure and streamlining the workflow process. The semi-robotic tracking method does not require an external tracking system or any disposables, reducing additional and recurrent costs of the biopsy procedure.
While the semi-robotic tracking method requires additional training for urologists to become acclimated to its mechanical arm, the learning curve has been estimated at as little as 10 patients.6 Companies like Focal Healthcare also offer hands-on and 24/7 remote support to help physicians easily and rapidly acquire the training they need.
Robotic arms have been criticized in the past for awkward movement with limited degrees of access; however, Focal Healthcare’s Fusion Bx device has significantly revitalized the robotic tracking method with its mechanical arm. The Fusion Bx’s mechanical arm has 6 degrees of freedom, allowing for an unrestricted range of motion and freehand-like access to the entire gland, utilizing the benefits of both mechanical and electromagnetic tracking without the limitations. Its patented counterbalance technology simplifies procedures by keeping the probe steady in any position, reducing the need for additional assistance. Lastly, the Fusion Bx’s arm moves with the patient to maintain coupling and minimize patient discomfort, which is often a source of patient movement.
What tracking method is best?
The field of fusion biopsy is rapidly evolving, and many different platforms have emerged onto the market. Each platform employs various technologies to achieve their results.
The addition of a MRI-US fusion biopsy device to a urological practice undoubtedly has many positive effects for both the practice and its patients; however, it also carries potential unseen financial, logistical, and procedural issues if not evaluated carefully prior to implementation. The tracking method employed by any fusion biopsy platform has a direct influence on the accuracy of the device, as well as on the logistical and financial operations of the practice that uses it.
Ultimately, the ideal tracking method in a fusion biopsy device will depend on the needs and preferences of each practice. Consideration of the many complex components of what makes a fusion biopsy device ideal for a practice should be taken into account to ensure that it can provide the best standard of care to its patients.
1 Siddiqui, M. M., Rais-Bahrami, S., Turkbey, B., George, A. K., Rothwax, J., Shakir, N., . . . Pinto, P. A. (2015). Comparison of MR/Ultrasound Fusion–Guided Biopsy With Ultrasound-Guided Biopsy for the Diagnosis of Prostate Cancer. JAMA, 313(4), 390–397. doi:10.1001/jama.2014.17942
2 Kongnyuy, M., George, A. K., Rastinehad, A. R., & Pinto, P. A. (2016). Magnetic Resonance Imaging-Ultrasound Fusion-Guided Prostate Biopsy: Review of Technology, Techniques, and Outcomes. Current Urology Reports, 17(4). doi:10.1007/s11934-016-0589-z
3 Tay, K. J., Gupta, R. T., Rastinehad, A. R., Tsivian, E., Freedland, S. J., Moul, J. W., & Polascik, T. J. (2016). Navigating MRI-TRUS fusion biopsy: Optimizing the process and avoiding technical pitfalls. Expert Review of Anticancer Therapy, 16(3), 303-311.doi:10.1586/14737140.2016.1131155
4 Maclachlan, R. A., Hollis, R. L., Martel, J. N., Lobes, L. A., & Riviere, C. N. (2017). Toward Improved Electromagnetic Tracking for Handheld Robotics. Proceedings of the 3rd International Conference on Mechatronics and Robotics Engineering – ICMRE 2017. doi:10.1145/3068796.3068823
5 Rothwax, J. T., George, A. K., Wood, B. J., & Pinto, P. A. (2014) Multiparametric MRI in Biopsy Guidance for Prostate Cancer: fusion-guided. BioMed Research International, 2014, 1-7. doi:10.1155/2014/439171
6 Kuru, T. H., Roethke, M. C., Seidenader, J., Simpfendörfer, T., Boxler, S., Alammar, K., . . . Hadaschik, B. A. (2013). Critical Evaluation of Magnetic Resonance Imaging Targeted, Transrectal Ultrasound Guided Transperineal Fusion Biopsy for Detection of Prostate Cancer. Journal of Urology, 190(4), 1380-1386. doi:10.1016/j.juro.2013.04.043