What causes prostate movement during a biopsy?
Prostate movement can be extremely difficult to determine because it can deform and dislocate independently from surrounding structures like the pubic bone and rectum.3 There are several points during the biopsy which can cause the prostate to move and/ or deform2, 4:
- The movement/ pressure from the probe can move and deform prostate
- Involuntary patient movement due to pain/ pressure from needle insertion
- Respiratory motion of the patient may shift the prostate
- Slow biopsy needle insertion in preparation for biopsy gun firing can deform the prostate
- Rapid biopsy needle insertion after firing the biopsy gun can cause deformation
What is an MRI-targeted fusion biopsy?
You’ve probably heard of magnetic resonance imaging (MRI). This high tech approach takes detailed images of the inner workings of your prostate. Prior to biopsy, these MR images are annotated with important information like the size and shape of your prostate, as well as any areas of suspicion. During biopsy, the physician will perform an ultrasound scan, or “sweep” of the prostate. Fusion software then fuses the ultrasound images with the annotated MR images by matching corresponding fiducial markers (set of marked locations on the prostate) and contours.1 This process is referred to as registration. The result is a reconstructed 3D model of the prostate with the suspected lesions.
What makes it “targeted”?
Spatial tracking of the probe, through means of mechanical or electromagnetic tracking, is crucial to targeting. In mechanical tracking, encoders in various joints track the probe and assign a global coordinate system to the ultrasound images. This allows the ultrasound probe position and projected needle path to be visualized relative to the 3D reconstruction. This tracking system is most effective when the target (prostate) remains static relative to the fiducial markers.2 Compared to the traditional method of “blind” sampling, where 10- 12 random cores are taken, this real-time tracking enables the operator to accurately navigate to specific targets in the prostate. This is not only useful for initial targeting, but also beneficial for re-sampling under active surveillance, and treatment planning.
How does prostate movement affect targeting?
Because it is difficult to determine the extent to which the prostate has moved and/ or deformed, it makes it equally difficult for the tracking system to account for it. Any differential between the ultrasound image and the 3D reconstruction results in a loss of accuracy in the MRI/ US fusion display. Consequently, this leads to inaccurate needle placement when using the fused display for targeted biopsies. If there is extreme movement, to the point the live ultrasound and 3D reconstruction are totally decoupled from one another, then the fused display will be unusable for targeting biopsies.2
How does the Fusion Bx address movement and deformation?
Focal Healthcare’s Fusion Bx system minimizes the potential for human error with its semi-robotic arm and automatic motion compensation. The key to maintaining accurate registration, or coupling, is to keep the prostate in the same shape and position as when the initial sweep was acquired. Equipped with patented counterbalance technology, our arm keeps the probe steady in any position, thus maintaining consistent pressure on the prostate and minimizing deformation. The 6 degrees of freedom and unrestricted range of motion means the probe can move with the patient, reducing patient discomfort.
Our motion compensation feature automatically accounts for potential prostate movement by tracking patient motion. Once the probe is positioned for targeting regions of interest and our semi-robotic arm is steady, this leaves the probe to move exclusively in the pitch (up and down) and yaw (side to side) directions. If the encoder values for the joints of the arm change, the system assumes the patient has moved and accounts for the difference.
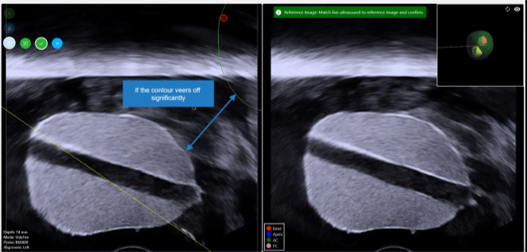
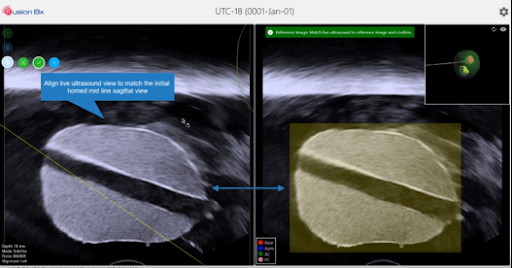
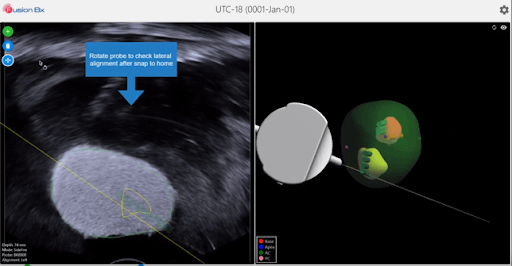
In the event of gross patient movement, and the ultrasound image becomes completely decoupled from the 3D construct (Fig 1), our “Snap to Home” feature can realign the images, allowing the procedure to continue within seconds. This feature enables the physician to manually adjust the probe until the live ultrasound matches the initial ultrasound “home” image – acquired during the sweep (Fig 2). Once satisfied with the result, the new coordinates replace the previous ones and the software realigns the 3D reconstruction with the “home” image. A quick sweep can ensure the ultrasound image matches the 3D construct (Fig 3).
Built around our 3 pillars, the Fusion Bx provides users with an accurate, efficient and accessible fusion biopsy solution.
1 Bjurlin, M. A., Rosenkrantz, A. B., & Taneja, S. S. (2017) MRI-fusion biopsy: the contemporary experience. Translational Andrology and Urology, 6(3), 483-489. doi:10.21037/tau.2017.04.30
2 Xu, S., Kruecker, J., Guion, P., Glossop, N., Neeman, Z., Choyke, P., … Wood, B. J. (2007). Closed-loop control in fused MR-TRUS image-guided prostate biopsy. Medical image computing and computer-assisted intervention : MICCAI … International Conference on Medical Image Computing and Computer-Assisted Intervention, 10(Pt 1), 128–135. doi:10.1007/978-3-540-75757-3_16
3 Xu, H., Lasso, A., Vikal, S., Guion, P., Krieger, A., Kaushal, A., … Fichtinger, G. (2010). MRI-guided robotic prostate biopsy: a clinical accuracy validation. Medical image computing and computer-assisted intervention : MICCAI … International Conference on Medical Image Computing and Computer-Assisted Intervention, 13(Pt 3), 383–391. doi:10.1007/978-3-642-15711-0_48
4 Silva, T. D., Fenster, A., Bax, J., Romagnoli, C., Izawa, J., Samarabandu, J., & Ward, A. D. (2011). Quantification of prostate deformation due to needle insertion during TRUS-guided biopsy: Comparison of hand-held and mechanically stabilized systems. Medical Physics, 38(3), 1718–1731. doi: 10.1118/1.3557883