Prostate cancer is the second most common and lethal cancer among men in the United States. In 2021, an estimated 250,000 men were diagnosed, with the majority over 65 years old. During the height of the pandemic, diagnosis and screening numbers plummeted, resulting in thousands of missed diagnoses. Lost in the surgical backlog, already delayed diagnoses may be further delayed, potentially lowering patient outcomes. That’s why early detection is crucial; for patients with local to regional prostate cancer (PCa), the 5 year survival rate is nearly 100%, but drops to 30% once the cancer begins to spread to other organs.1 The current standard diagnostic pathway is a PSA test followed by a systematic biopsy if the results are positive. The PSA test is a highly sensitive biomarker, meaning it can yield a lot of false negatives and even more false positives. The sensitivity and specificity of the PSA test is so poor that recommendations have been made to discontinue prostate cancer screening in the US.2 Still, it’s not totally useless, PSA testing is associated with 20% reduction in PCa- specific mortality, but sometimes it may do more harm than good.3 Unlike mpMRI, the PSA test cannot differentiate between indolent and lethal cancer. Consequently, many men are subject to the psychological, economic and physical stresses from unnecessary biopsies, potential infection and overtreatment. Eliminating screening spares the patient from overdiagnosis, but can result in late detection of intermediate and high grade cancers. Either way, a confirmatory biopsy is required for definitive diagnosis.4
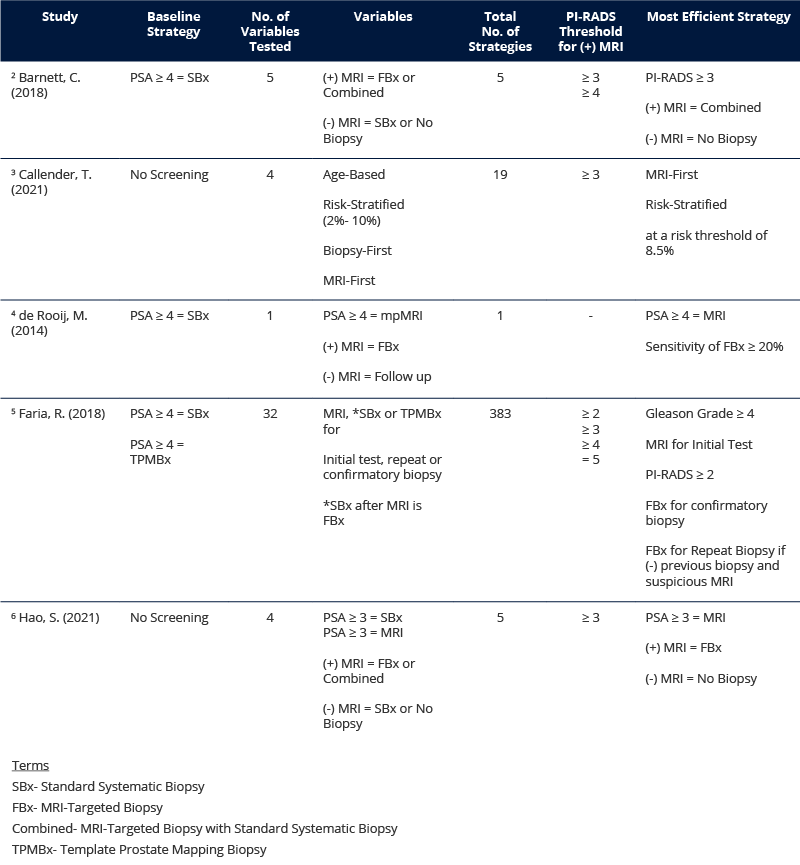
Effective screening requires better pretreatment characterization of PCa to ensure that the benefits of diagnosis outweigh the risks.4 MRI is an alternative tool to the PSA test. It is highly accurate, minimally invasive and reduces the risk of overtreatment. Increasingly, it is recommended for diagnosis if the initial biopsy is negative, but could be potentially more valuable as a triage test, deciding who needs a biopsy and how it may best be conducted.5 The EAU (European Association of Urology) recommends the use of MRI for both biopsy naive and previously negative patients, where men with PI-RADS ≥ 3 undergo combined targeted and systematic biopsy. Similarly, Swedish guidelines recommend MRI for both biopsy naive and previously negative patients, but a positive MRI should be followed by targeted biopsy only.6 Regardless of the fine print, an MRI pathway can reduce unnecessary biopsies, overtreatment and associated costs by 30- 60%.3, 4, 6 Several existing studies show that compared to the standard PSA test and systematic biopsy, an MRI-first strategy can be cost effective while also improving the patient’s quality of life (Table 1 see below).
A 2009 cross European study found that PCa had the fourth highest cancer cost, making up 7% of all cancer costs for a total of €8.43 billion. If 40% of biopsies, and subsequent treatment, are unnecessary, that means up to 40% of all PCa costs are unnecessary. That’s roughly €3.4 billion!6 Using an MRI-first strategy may mean additional costs associated with MRI scans, but these can be offset by the reduction in biopsies, biopsy cores and over treatment.2, 3, 4,5, 6 In the Callender study, MRI-first biopsies resulted in 0.9% fewer deaths from cancer, 14.9% fewer overdiagnosis and 33.8% fewer biopsies. It led to a 4.8-fold increase in MRI scans, but the overall cost was actually lower than a biopsy-first approach.3 The de Rooij study also found the initial costs were compensated for by the reduction in treatment costs due to fewer false positives and improved characterization of tumor aggressiveness. Even before accounting for biopsy needles or post biopsy complications, the MRI strategy cost was almost equal with the current standard of care. MRI also led to an improved quality of life by avoiding unnecessary radical treatment of insignificant tumors and decreasing the likelihood of detecting significant PCa later.4 Findings from the Hao study showed standard systematic biopsy (SBx) was overall more costly and less effective than either of the MRI-first strategies. MRI-targeted biopsy (FBx) alone was considered the optimal choice for Sweden since the combined biopsy had a high incremental cost for a low incremental gain in quality of life.6 Whether performing a targeted biopsy or combined following a positive MRI, studies show that performing SBx on a negative MRI is more costly and less effective than performing no biopsy at all.2, 6 Compared to the biopsy-first standard, taking an MRI-first pathway, like the fusion biopsy workflow (Fig.1), is associated with a greater quality of life that can be achieved at reduced costs.3, 4, 6
The fusion biopsy workflow with the Fusion Bx uses MRI as a triage test, where negative results avoid biopsy and positive results are used for targeting. With key features like real-time spatial tracking of the probe, freehand-like range of motion and minimized prostate deformation, the Fusion Bx enables physicians to accurately and consistently perform MRI-targeted biopsies using minimal cores. Supporting both transrectal and transperineal (both traditional and freehand) approaches, the same 4 step guided workflow is easy to follow, which means quicker training. The Fusion Bx is a cost effective and easy-to-learn system that helps make MRI-targeted biopsies more accessible to physicians and patients. By adopting an MRI-first fusion pathway, physicians can get meaningful results faster to provide better treatment, saving money and ultimately, providing patients with peace of mind.
1 Panzone, J., Byler, T., Bratslavsky, G., Goldberg, H. (2022). Transrectal Ultrasound in Prostate Cancer: Current Utilization, Integration with mpMRI, HIFU and Other Emerging Applications. Cancer Management and Research, 2022(14), 1209- 1228. https://doi.org/10.2147/CMAR.S265058
2 Barnett, C. L., Davenport, M. S., Montgomery, J. S., Wei, J. T., Montie, J. E., & Denton, B. T. (2018). Cost‐effectiveness of magnetic resonance imaging and targeted fusion biopsy for early detection of prostate cancer. BJU International, 122(1), 50–58. https://doi.org/10.1111/bju.14151
3 Callender, T., Emberton, M., Morris, S., Pharoah, P. D. P., Pashayan, N. (2021). Benefit, Harm, and Cost-effectiveness Associated With Magnetic Resonance Imaging Before Biopsy in Age-based and Risk-stratified Screening for Prostate Cancer. JAMA Network Open, 4(3):e2037657. doi:10.1001/jamanetworkopen.2020.37657
4 de Rooij, M., Crienen, S., Witjes, J. A., Barentsz, J. O., Rovers, M. M., & Grutters, J. P. (2014). Cost-effectiveness of magnetic resonance (MR) imaging and MR-guided targeted biopsy versus systematic transrectal ultrasound-guided biopsy in diagnosing prostate cancer: a modelling study from a health care perspective. European Urology, 66(3), 430–436. https://doi.org/10.1016/j.eururo.2013.12.012
5 Faria, R., Soares, O. M., Spackman, E., Ahmed, U. H., Brown, C. L., Kaplan, R., Emberton, M., & Sculpher, J. M. (2018). Optimising the Diagnosis of Prostate Cancer in the Era of Multiparametric Magnetic Resonance Imaging: A Cost-effectiveness Analysis Based on the Prostate MR Imaging Study (PROMIS). European Urology, 73(1), 23- 30. https://doi.org/10.1016/j.eururo.2017.08.018.
6 Hao, S., Karlsson, A., Heintz, E., Elfstrom Miriam, K., Nordström, T., & Clements, M. (2021). Cost-Effectiveness of Magnetic Resonance Imaging in Prostate Cancer Screening: A Microsimulation Study. Value in Health, 24(12), 1763- 17772. https://doi.org/10.1016/j.jval.2021.06.001